Quality By Design (QBD) Approach in Processing of Nanoparticles Loading Antifungal Drugs
Main Article Content
Abstract
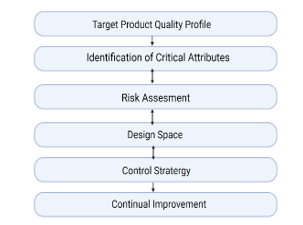
"Quality by Design" (QbD) is a strategic process for development and manufacturing, it is aimed to make sure that a final pharmaceutical product performs as expected, both in terms of purity and efficacy. To achieve this, QbD implementation in the manufacturing process must be done clearly with defined objectives and proper risk management. Considering that it provides drug developers with a better understanding of manufacturing processes, reduces the frequency of batch failures, and therefore is data-driven, the QbD methodology contributes to the progressive manufacturing environment and provides a higher return on investment. Applying QbD in nano formulation can greatly impact advanced drug delivery. Particles with a diameter of less than 100 nm are known as nanoparticles, are extensively studied in nanotechnology, and have wide application in novel drug delivery systems, targeted sites, and dosing regimens. Controlling particle size, surface characteristics, and the release of pharmacologically active substances are the main criteria when formulating nanoparticles as a delivery system. As fungal infections are increasing rapidly which affect the skin, hair, nails, or mucous membranes and can also infect the lungs or other parts of your body, implementing QbD in the processing of nanoparticles loading antifungals might be of great need.
Article Details
References
Kim JY. Human fungal pathogens: Why should we learn? J Microbiol. 2016;54(3):145–8.
Lockhart SR, Diekema DJ, Pfaller MA. The epidemiology of fungal infections. Clin Mycol with CD-ROM. 2009;1–14.
Rudramurthy SM, Shaw D. Epidemiology of superficial fungal infections in Asia. Clin Pract Med Mycol Asia. 2019;9–37.
Vallabhaneni S, Mody RK, Walker T, Chiller T. The Global Burden of Fungal Diseases. Infect Dis Clin North Am. 2016;30(1):1–11.
Nett, J.E., Andes DR. Antifungal agents: spectrum of activity, pharmacology, and clinical indications.
Lewis RE. Current concepts in antifungal pharmacology. Mayo Clin Proc. 2011;86(8):805–17.
Zazo H, Colino CI, Lanao JM. Current applications of nanoparticles in infectious diseases. J Control Release. 2016;224:86–102.
Roco MC. Broader societal issues of nanotechnology. J Nanoparticle Res. 2003;5(3–4):181–9.
Saltzman W. Drug delivery: engineering principles for drug therapy. Drug Deliv Eng Princ Drug Ther [Internet]. 2001;235–63. Available from: http://books.google.com/books?hl=en&lr=&id=Z5UDv8pCprUC&oi=fnd&pg=PA1&dq=Drug+Delivery:+Engineering+Principles+for+Drug+Therapy&ots=luKr8IJeCk&sig=jOin-nsVBzl2c89z6Yj6uWGnGFM
Vila A, Sánchez A, Tobío M, Calvo P, Alonso MJ. Design of biodegradable particles for protein delivery. J Control Release. 2002;78(1–3):15–24.
Mu L, Feng SS. A novel controlled release formulation for the anticancer drug paclitaxel (taxol ®): PLGA nanoparticles containing vitamin E TPGS. Chemother Eng Collect Pap Si-Shen Feng - A Tribut to Shu Chien His 82nd Birthd. 2013;246–305.
Rathore AS, Winkle H. Quality by design for biopharmaceuticals. Nat Biotechnol. 2009;27(1):26–34.
Yu LX, Amidon G, Khan MA, Hoag SW, Polli J, Raju GK, et al. Understanding pharmaceutical quality by design. AAPS J. 2014;16(4):771–83.
Charoo NA, Shamsher AAA, Zidan AS, Rahman Z. Quality by design approach for formulation development: A case study of dispersible tablets. Int J Pharm. 2012;423(2):167–78.
Soni G, Yadav KS, Gupta MK. Design of Experiments (DoE) Approach to Optimize the Sustained Release Microparticles of Gefitinib. Curr Drug Deliv. 2018;16(4):364–74.
Leuenberger H, Puchkov M, Krausbauer E, Betz G. Manufacturing pharmaceutical granules: Is the granulation end-point a myth? Powder Technol. 2009;189(2):141–8.
Miller CE. Chemometrics and NIR: A match made in heaven,.
Incecayir T, Sun J, Tsume Y, Xu H, Gose T, Nakanishi T, et al. Carrier-Mediated Prodrug Uptake to Improve the Oral Bioavailability of Polar Drugs: An Application to an Oseltamivir Analogue. J Pharm Sci. 2016;105(2):925–34.
Belasco JM. Target product profile: beginning drug development with the end in mind. Udate Rep. 2007;1:36–9.
Nahar M, Dutta T, Murugesan S, Asthana A, Mishra D, Rajkumar V, et al. Functional polymeric nanoparticles: An efficient and promising tool for active delivery of bioactives. Crit Rev Ther Drug Carrier Syst. 2006;23(4):259–318.
Farokhzad OC, Langer R. Impact of nanotechnology on drug delivery. ACS Nano. 2009;3(1):16–20.
García-Corvillo MDP. Polymeric nanoparticles for drug delivery to the central nervous system via nasal route. Ars Pharm. 2016;57(1):27–35.
Lin G, Zhang H, Huang L. Smart polymeric nanoparticles for cancer gene delivery. Mol Pharm. 2015;12(2):314–21.
Zhong Y, Meng F, Deng C, Zhong Z. Ligand-directed active tumor-targeting polymeric nanoparticles for cancer chemotherapy. Biomacromolecules. 2014;15(6):1955–69.
Kamaly N, Xiao Z, Valencia PM, Radovic-Moreno AF, Farokhzad OC. Targeted polymeric therapeutic nanoparticles: Design, development and clinical translation. Chem Soc Rev. 2012;41(7):2971–3010.
Krasia-Christoforou T, Georgiou TK. Polymeric theranostics: Using polymer-based systems for simultaneous imaging and therapy. J Mater Chem B. 2013;1(24):3002–25.
Schaffazick SR, Pohlmann AR, Dalla-Costa T, Guterres SS. Freeze-drying polymeric colloidal suspensions: Nanocapsules, nanospheres and nanodispersion. A comparative study. Eur J Pharm Biopharm. 2003;56(3):501–5.
Crucho CIC, Barros MT. Polymeric nanoparticles: A study on the preparation variables and characterization methods. Mater Sci Eng C. 2017;80:771–84.
Guterres SS, Alves MP, Pohlmann AR. Polymeric Nanoparticles, Nanospheres and Nanocapsules, for Cutaneous Applications. Drug Target Insights. 2007;2:117739280700200.
Christoforidis JB, Chang S, Jiang A, Wang J, Cebulla CM. Intravitreal devices for the treatment of vitreous inflammation. Mediators Inflamm. 2012;2012.
Nagavarma BVN, Yadav HKS, Ayaz A, Vasudha LS, Shivakumar HG. Different techniques for preparation of polymeric nanoparticles- A review. Asian J Pharm Clin Res. 2012;5(SUPPL. 3):16–23.
Mora-Huertas CE, Fessi H, Elaissari A. Polymer-based nanocapsules for drug delivery. Int J Pharm. 2010;385(1–2):113–42.
Ezhilarasi PN, Karthik P, Chhanwal N, Anandharamakrishnan C. Nanoencapsulation Techniques for Food Bioactive Components: A Review. Food Bioprocess Technol. 2013;6(3):628–47.
Charrueau C, Zandanel C. Drug Delivery by Polymer Nanoparticles: The Challenge of Controlled Release and Evaluation. Polym Nanoparticles Nanomedicines. 2016;439–503.
Nicolas J, Mura S, Brambilla D, Mackiewicz N, Couvreur P. Design, functionalization strategies and biomedical applications of targeted biodegradable/biocompatible polymer-based nanocarriers for drug delivery. Chem Soc Rev. 2013;42(3):1147–235.
Odds, F.C.; Brown, A.J.; Gow N. Antifungal agents: Mechanisms of action.
ALrawashdeh I, Qaralleh H, Al-limoun M, Khleifat K. Antibactrial Activity of Asteriscus graveolens Methanolic Extract: Synergistic Effect with Fungal Mediated Nanoparticles against Some Enteric Bacterial Human Pathogens. J Basic Appl Res Biomed. 2019;5(2):89–98.
Vera-González N, Bailey-Hytholt CM, Langlois L, de Camargo Ribeiro F, de Souza Santos EL, Junqueira JC, et al. Anidulafungin liposome nanoparticles exhibit antifungal activity against planktonic and biofilm Candida albicans. J Biomed Mater Res - Part A. 2020;108(11):2263–76.
Mahmood A, Rapalli VK, Waghule T, Gorantla S, Singhvi G. Luliconazole loaded lyotropic liquid crystalline nanoparticles for topical delivery: QbD driven optimization, in-vitro characterization and dermatokinetic assessment. Chem Phys Lipids. 2021;234.
Spósito PÁ, Mazzeti AL, Faria C de O, Aurbina J, Pound-Lana G, Bahia MT, et al. Ravuconazole self-emulsifying delivery stem: In vitro activity against Trypanosoma cruzi amastigotes and in vivo toxicity. Int J Nanomedicine. 2017;12:3785–99.
Liu M, Chen M, Yang Z. Design of amphotericin B oral formulation for antifungal therapy. Drug Deliv. 2017;24(1):1–9.
Odds, F.C.; Brown, A.J.; Gow NA. Antifungal agents: Mechanisms of action.
Janknegt R, de Marie S, Bakker-Woudenberg IAJM, Crommelin DJA. Liposomal and Lipid Formulations of Amphotericin B: Clinical Pharmacokinetics. Clin Pharmacokinet. 1992;23(4):279–91.
Vermes A, Guchelaar HJ, Dankert J. Flucytosine: A review of its pharmacology, clinical indications, pharmacokinetics, toxicity and drug interactions. J Antimicrob Chemother. 2000;46(2):171–9.
Ashe, W.D. J. VR. D.E. 5-fluorocytosine: A brief review.
Larsen RAF. In Essentials of Clinical Mycology; Kauffman, C.A., Pappas, P.G., Sobel, J.D., Dismukes, W.E.,.
Viviani MA. Flucytosine—What is its future?
Chang YL, Yu SJ, Heitman J, Wellington M, Chen YL. New facets of antifungal therapy. Virulence. 2017;8(2):222–36.
Pianalto KM, Alspaugh JA. New horizons in antifungal therapy. J Fungi. 2016;2(4).
Martínez-Matías N, Rodríguez-Medina JR. Fundamental concepts of azole compounds and triazole antifungals: A beginner’s review. P R Health Sci J. 2018;37(3):135–42.
Peyton, L.R.; Gallagher S. H. M. Triazole antifungals: A review.
Cowen LE, Sanglard D, Howard SJ, Rogers PD, Perlin DS. Mechanisms of antifungal drug resistance. Cold Spring Harb Perspect Med. 2015;5(7).
Cappelletty D, Eiselstein-McKitrick K. The echinocandins. Pharmacotherapy. 2007;27(3):369–88.
Johnson MD, Perfect JR. Caspofungin: First approved agent in a new class of antifungals. Expert Opin Pharmacother. 2003;4(5):807–23.
Chen SCA, Slavin MA, Sorrell TC. Echinocandin antifungal drugs in fungal infections: A comparison. Drugs. 2011;71(1):11–41.
Sucher AJ, Chahine EB, Balcer HE. Echinocandins: The newest class of antifungals. Ann Pharmacother. 2009;43(10):1647–57.
Patil A, Majumdar S. Echinocandins in antifungal pharmacotherapy. J Pharm Pharmacol. 2017;69(12):1635–60.
Ia IA. Practice guidelines for the diagnosis and management of aspergillosis. 2021;62(10):1282–7.
Aigner M, Lass-Flörl C. Treatment of drug-resistant Aspergillus infection. Expert Opin Pharmacother. 2015;16(15):2267–70.
Te Welscher YM, Van Leeuwen MR, De Kruijff B, Dijksterhuis J, Breukink E. Polyene antibiotic that inhibits membrane transport proteins. Proc Natl Acad Sci U S A. 2012;109(28):11156–9.
Anderson TM, Clay MC, Cioffi AG, Diaz KA, Hisao GS, Tuttle MD, et al. Amphotericin forms an extramembranous and fungicidal sterol sponge. Nat Chem Biol. 2014;10(5):400–6.
Gray KC, Palacios DS, Dailey I, Endo MM, Uno BE, Wilcock BC, et al. Amphotericin primarily kills yeast by simply binding ergosterol. Proc Natl Acad Sci U S A. 2012;109(7):2234–9.
Hope WW, Tabernero L, Denning DW, Anderson MJ. Molecular mechanisms of primary resistance to flucytosine in Candida albicans. Antimicrob Agents Chemother. 2004;48(11):4377–86.
Whaley SG, Berkow EL, Rybak JM, Nishimoto AT, Barker KS, Rogers PD. Azole antifungal resistance in Candida albicans and emerging non-albicans Candida Species. Front Microbiol. 2017;7(JAN).
Nayak AK, Ahmad SA, Beg S, Ara TJ, Hasnain MS. Drug delivery: Present, past, and future of medicine. Appl Nanocomposite Mater Drug Deliv. 2018;255–82.
Singhvi G, Rapalli VK, Nagpal S, Dubey SK, Saha RN. Nanocarriers as Potential Targeted Drug Delivery for Cancer Therapy. 2020;51–88.
Yu LX. Pharmaceutical quality by design: Product and process development, understanding, and control (Pharmaceutical Research DOI: 10.1007/s11095-007- 9511-1). Pharm Res. 2008;25(10):2463.
Falusi F, Budai-Szűcs M, Csányi E, Berkó S, Spaits T, Csóka I, et al. Investigation of the effect of polymers on dermal foam properties using the QbD approach. Eur J Pharm Sci. 2022;173.
Lipner SR, Scher RK. Onychomycosis: Treatment and prevention of recurrence. J Am Acad Dermatol. 2019;80(4):853–67.